
Biotech Brand Strategy & Market Entry FAQs | Life Sciences Brand Development Explained
Introduction
The biotechnology market is projected to reach $2.42 trillion in 2026, growing at a 12.67% CAGR through 2031, yet the commercial success of a biotech asset is no longer determined by science alone. In a capital environment where investors now demand commercial clarity as early as Phase 2, and where acquirers and strategic partners evaluate brand narrative alongside clinical data, biotech companies face a challenge that many are underprepared for: building a credible, differentiated brand before a product has received regulatory approval. This article demystifies biotech brand strategy and market entry, covering what it means to build a life sciences brand, why it must begin earlier than most organizations expect, and how the decisions made in pre-commercial stages determine competitive position at and after launch.
This FAQ is written for commercial leads, heads of marketing, CEOs, and chief business officers at clinical-stage and emerging biotech companies who are evaluating how to develop a brand strategy that works simultaneously for multiple demanding audiences: investors, healthcare professionals (HCPs), payers, patients, and potential strategic partners. By the end, the reader will have a clear framework for understanding what biotech brand strategy requires, when to start, and what distinguishes the brands that build lasting market presence from those that rely on science alone.
Market Context: Disruption & Opportunity
Biotech enters 2026 in a disciplined capital environment where the rules for commercial success have fundamentally shifted. After years of volatility, investor sentiment is improving, biotech venture financing rose approximately 71% quarter-over-quarter to $3.1 billion in Q3 2025, and M&A activity is accelerating as large pharma addresses a looming patent cliff that puts over 40% of big pharma revenue at risk from patent expirations in the next six years. That M&A pressure is creating significant commercial opportunity for biotech brands with late-stage pipelines and clean IP, but only for those that can tell a compelling commercial story alongside their clinical data.
The implication for biotech brand strategy is direct: acquirers and investors now demand commercial clarity earlier in clinical development than ever before. Phase 2 readouts that once secured financing now require accompanying evidence of market access strategy, competitive positioning, and launch economics. Clinical differentiation alone will not overcome structural positioning disadvantages, a reality that makes brand strategy not a post-approval consideration but a pre-clinical investment. For biotech commercial teams, this shift creates both urgency and competitive opportunity: companies that build robust brand strategy infrastructure early are consistently better positioned for fundraising, partnerships, and launch success than those that treat branding as a communications activity bolted onto the end of the development process.
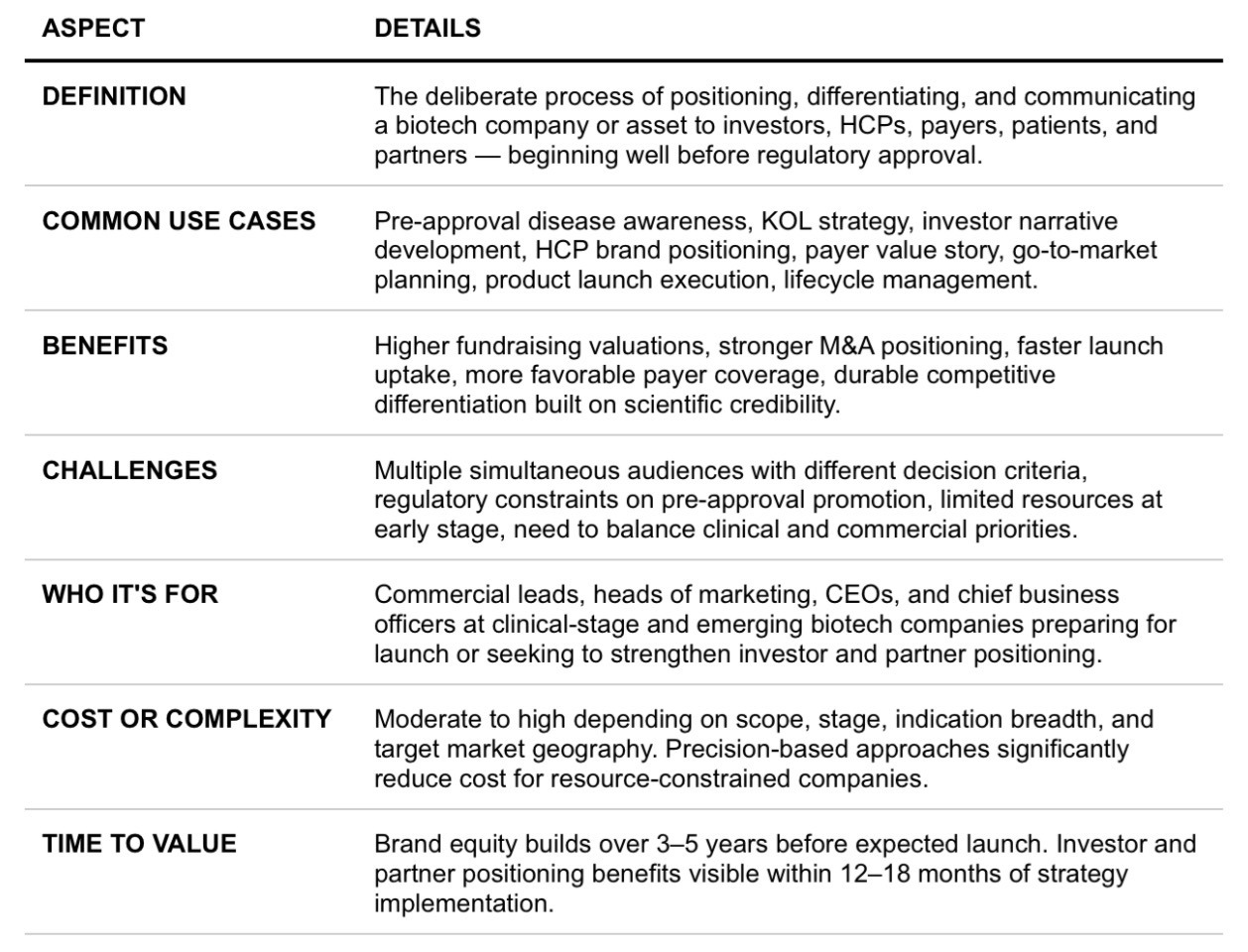
FAQs Snapshot

The following questions represent the most common and strategically important queries biotech commercial leaders raise when evaluating brand strategy and market entry planning.
What is biotech brand strategy?
Biotech brand strategy is the deliberate process of defining how a company or asset is positioned, differentiated, and communicated to the multiple stakeholder audiences, investors, HCPs, payers, patients, and potential partners, that determine its commercial success. Unlike consumer brand strategy, which is primarily aimed at a single buyer audience, biotech brand strategy must simultaneously address the concerns of scientific credibility (for HCPs and regulators), financial viability (for investors), health economic value (for payers), and emotional accessibility (for patients and caregivers). A well-constructed biotech brand strategy aligns all of these messages within a coherent narrative architecture, one that communicates the scientific evidence clearly, translates it into outcomes that each audience cares about, and builds the trust and credibility required to drive prescribing, coverage, investment, and partnership decisions.
When should a biotech company start building its brand?
The commercially successful answer is: earlier than most companies do. Many biotech companies treat brand development as a pre-launch activity, something to be addressed in the twelve to eighteen months before regulatory approval. In practice, the companies that achieve the strongest launch performance begin brand strategy work during Phase 2 clinical development, when there is enough clinical data to define a credible value proposition but enough time to shape market perceptions, engage KOLs, and build the disease awareness infrastructure that accelerates HCP and patient identification at launch. Investors now specifically evaluate commercial readiness as part of funding decisions, meaning that a clearly articulated brand strategy, including target patient archetype, competitive positioning, and payer value narrative, directly influences a company's ability to raise capital and attract strategic partners before approval.
What is the difference between a biotech brand and a product brand?
A biotech corporate brand represents the company as a whole, its scientific platform, leadership credibility, innovation philosophy, and track record, and is the primary currency in investor, partner, and talent relationships. A product brand is specific to an individual therapeutic asset, its clinical profile, patient population, HCP positioning, and market access narrative. In the earliest stages of development, the corporate brand carries nearly all of the commercial weight, since no approved product yet exists. As a company moves toward approval, product brand strategy becomes increasingly important, and the two must be designed to reinforce each other: a strong corporate brand lends credibility to product claims, while successful product launches build the corporate brand's commercial reputation. For biotech companies managing multiple pipeline assets, brand architecture, how corporate and product brands relate to each other, becomes a strategic decision that affects investor perception, HCP awareness, and payer engagement simultaneously.
How do you build a biotech brand before FDA approval?
Pre-approval brand building operates through a specific set of activities that do not require approved promotional claims. Disease awareness programs establish the unmet need that a company's asset is designed to address, building the HCP and patient identification infrastructure that accelerates launch uptake. KOL and key opinion leader strategy involves engaging the scientific and clinical thought leaders whose publication activity, congress presentations, and peer relationships shape how a therapeutic area is understood and how new treatments are evaluated. Scientific communication, through publications, data presentations at major congresses, and medical education content, establishes clinical credibility for the science and the team behind it. Investor communications build the commercial narrative that frames the asset's market opportunity, competitive differentiation, and value hypothesis in terms that resonate with the financial audiences that fund development. Together, these activities build the brand equity that transforms a clinical data package into a commercially anticipated launch.
What audiences does a biotech brand strategy need to address?
Biotech brand strategy must address at least four distinct audience segments, each with fundamentally different decision criteria. Healthcare professionals evaluate scientific evidence, safety and efficacy versus standard of care, mechanism of action clarity, and practical prescribing and administration factors. Payers evaluate health economic value, cost per quality-adjusted life year, budget impact, and comparative effectiveness versus existing treatments, and require a value narrative that goes beyond clinical data to demonstrate economic justification for coverage and reimbursement. Investors and strategic partners evaluate market opportunity size, competitive differentiation, regulatory pathway risk, commercialization feasibility, and team credibility. Patients and patient advocacy organizations evaluate clinical benefit in real-world terms, access and affordability, and the degree to which a company demonstrates genuine understanding of the patient experience. Effective biotech brand strategy does not use a single message for all of these audiences, it uses a coherent underlying narrative that is translated into audience-specific value propositions, each addressing the specific concerns and decision criteria of that stakeholder group.
What is a target product profile (TPP) and how does it relate to brand strategy?
A target product profile (TPP) is a clinical and regulatory document that defines the desired attributes of a product, intended indication, efficacy endpoints, safety profile, dosing, and route of administration, used to guide clinical development decisions. Its relationship to brand strategy is direct and underappreciated: the TPP defines the clinical claims that will be available at launch, which in turn determines the brand's ability to differentiate from existing treatments and justify its pricing and access strategy. Biotech companies that engage commercial and brand strategy teams in TPP development, before the document is finalized, consistently produce label language that is more commercially useful, more differentiated in payer evaluations, and more clearly positioned against the competitive landscape than those that treat TPP as a purely clinical activity. Integrating commercial perspective into TPP development is one of the highest-value interventions available to clinical-stage biotech companies.
How does biotech go-to-market strategy differ from pharma go-to-market strategy?
The fundamental difference lies in resources, speed, and audience complexity. Large pharmaceutical companies launch with established commercial infrastructure, large sales forces, deep payer relationships, and brand recognition built over decades. Biotech companies, particularly first-time launchers, must build commercial capability from scratch, often while simultaneously completing late-stage clinical development and managing investor expectations. This constraint demands greater commercial precision: rather than the broad promotional investment that established pharma can sustain, biotech go-to-market strategy must prioritize the highest-value HCP segments, the most tractable payer pathways, and the most efficient channels for reaching each audience. Data-driven approaches, AI-enabled next-best-action models for HCP prioritization, precision digital channels for patient identification, and modular content strategies for multi-stakeholder engagement, are increasingly the default for biotech commercial teams that need to maximize impact with limited resources.
What role do KOLs play in biotech brand strategy?
Key opinion leaders, the physicians, researchers, and academic clinicians whose expertise shapes how a therapeutic area is understood and how new treatments are evaluated, are among the most valuable assets in a biotech brand strategy. KOL engagement serves multiple strategic functions: it generates the scientific publications, congress presentations, and clinical guidelines that establish clinical credibility for a new mechanism or patient population; it builds the peer-to-peer awareness that accelerates HCP adoption at launch; and it provides the company with real-world clinical insight that strengthens both the clinical development program and the commercial value proposition. Effective KOL strategy begins two to three years before anticipated launch, identifying the right scientific and clinical leaders, co-creating educational and publication opportunities, and building the advisory relationships that make KOLs genuine advocates rather than transactional consultants.
How do payer strategy and brand strategy intersect in biotech?
Payer strategy and brand strategy are inseparable in biotech because coverage and reimbursement decisions determine whether a drug is accessible to the patients who need it, and therefore whether launch uptake matches clinical expectations. The brand's value narrative must be designed from the beginning with payer needs in mind: health economic outcomes data, budget impact modeling, and comparative effectiveness evidence are not additions to a clinical story but core components of the brand's value proposition. Biotech companies that engage payers early, in advisory capacities during late-stage development, through AMCP dossier development, and through market access strategy built into the brand architecture, consistently achieve faster coverage, more favorable formulary positioning, and higher launch uptake than those that address payer strategy as a post-approval problem. In an environment of intensifying pricing scrutiny, the biotech brands that frame their value in health economic terms from the earliest stages of commercial development are the ones that survive payer negotiations with their pricing and access strategy intact.
What metrics matter most for evaluating biotech brand strategy performance?
The metrics that matter most align with the stage of development and the primary strategic objective. In pre-commercial stages, the relevant indicators are investor sentiment measures, valuation multiples, term sheet quality, and strategic partner interest, alongside KOL engagement depth, disease awareness reach among target HCP segments, and share of scientific voice at major congresses. As a company approaches launch, payer-facing metrics become critical: formulary win rates, coverage decision timelines, and access restriction rates relative to competitive benchmarks. Post-launch, the standard commercial performance indicators, peak sales trajectory, HCP prescribing adoption curves, patient identification rates, and market share versus competitive products, become the primary measures of brand strategy effectiveness. Throughout all stages, brand tracking research measuring aided and unaided awareness, message recall, and attribute association among priority HCP and investor audiences provides the leading indicator data that allows commercial teams to adjust strategy before performance gaps become launch failures.
How long does it take to build a biotech brand?
Meaningful biotech brand equity cannot be built quickly, the credibility and awareness that drive commercial success are accumulated over years, not months. A realistic timeline for a clinical-stage company investing in brand strategy from Phase 2 onward spans three to five years before the expected launch date. The first phase, typically eighteen to twenty-four months, establishes the scientific and corporate brand foundations: disease awareness, KOL engagement, publication strategy, and investor communications architecture. The second phase, running through Phase 3 and the regulatory submission period, develops the HCP-facing brand, refines the payer value narrative based on emerging clinical data, and builds the commercial infrastructure required for launch. The launch period itself, typically the twelve months surrounding approval and initial commercialization, executes the brand strategy at full commercial scale. Companies that compress this timeline by beginning brand strategy work later consistently experience the same outcome: launch uptake that underperforms the clinical opportunity because the market was not adequately prepared.
Can a small biotech company with limited resources build an effective brand strategy?
Resource constraints are a reality for most clinical-stage biotech companies, and they are not an insurmountable barrier to effective brand strategy, but they do require a different approach than large pharma commercial playbooks provide. The most effective approach for resource-constrained biotechs is precision over breadth: identifying the narrowest possible HCP segment that represents the highest-value prescribers for the target indication, designing a message architecture that can be communicated through digital channels with limited sales force support, and prioritizing the two or three payer accounts whose coverage decisions will most influence launch uptake. Data-driven precision, using AI tools for HCP prioritization, digital channels for stakeholder education, and modular content frameworks for multi-audience communication, enables first-time launchers to achieve commercial impact that previously required significantly larger investments. The companies that succeed commercially despite resource constraints are consistently those that define their beachhead precisely, execute with discipline, and avoid the trap of trying to replicate large-pharma commercial models with early-stage budgets.
Benefits of a Biotech Brand Strategy & Market Entry Program
A well-executed biotech brand strategy and market entry program delivers value that extends far beyond launch performance. The most immediate benefit is fundraising advantage: companies with a clearly articulated brand narrative, defined target patient population, and credible commercial strategy consistently command higher valuations and more favorable term sheets than those presenting clinical data without commercial context. In the M&A environment of 2026, where acquirers are specifically targeting assets with clean IP, late-stage data, and commercial clarity, a mature brand strategy directly increases asset value and acquisition competitiveness. Beyond financing, pre-commercial brand building accelerates launch uptake by ensuring that the HCPs, patients, and payers who need to act on approval are already aware, educated, and engaged rather than encountering the therapy for the first time at launch. The compounding benefit is market position durability: brands that establish scientific credibility and stakeholder relationships early are significantly harder for competitive entrants to displace than those that rely on post-approval promotional investment alone.
Quick Summary Table
Let’s kickstart the conversation and design stuff people will love.

Deep-Dive Sections

What Biotech Brand Strategy Is and Why It Matters
Biotech brand strategy is not a communications exercise, it is a commercial infrastructure decision that determines whether a company can translate scientific innovation into market value. Its strategic importance in 2026 is driven by a fundamental change in how investors, acquirers, and partners evaluate biotech assets: clinical data is necessary but no longer sufficient. The question acquirers are asking is not just whether the science works but whether the brand can win, whether the patient population is clearly defined, the competitive differentiation is credible and defensible, the payer value story will survive reimbursement review, and the HCP audience is being educated and prepared in advance of launch. Companies that answer these questions with a mature brand strategy consistently achieve better financial outcomes, higher valuations, more favorable partnership terms, and stronger launch performance, than those that treat commercial development as a post-approval activity.
How Biotech Brand Strategy and Clinical Development Work Together
The most commercially effective biotech organizations design brand strategy and clinical development as integrated, not sequential, activities. Clinical trial design decisions, endpoint selection, patient population definition, comparator arm choices, safety monitoring thresholds, have direct brand strategy implications that are often not recognized until the label is received and commercial constraints become apparent. Companies that engage commercial and brand strategy perspectives during protocol design consistently produce trial results that are more differentiating in competitive contexts, more compelling in payer evaluations, and more actionable in HCP communications than those that treat clinical and commercial as separate workstreams. The integration point is the target product profile: when the TPP is developed with commercial input, it encodes the brand's competitive differentiation into the clinical development program itself, creating alignment between the science and the commercial story that cannot be retrofitted after the data is in.
When to Invest in Corporate Brand vs. Product Brand
The sequencing of corporate and product brand investment is one of the most important and least discussed decisions in biotech commercial strategy. In the earliest stages of company development, the corporate brand carries the full commercial weight: it is what attracts investors, recruits scientific talent, secures partnership conversations, and builds the institutional credibility that accelerates regulatory and payer engagement. Corporate brand investment at this stage means defining a clear platform narrative, building scientific credibility through publication and congress presence, and developing the investor communications architecture that positions the company's pipeline in the context of market opportunity and strategic value. Product brand investment should begin at or before Phase 2 initiation, when the asset's clinical profile is sufficiently defined to support a differentiated positioning hypothesis. The two must be designed to reinforce each other, a strong corporate brand narrative that encompasses the product's clinical innovation creates a halo effect that benefits individual asset valuations, while product successes compound corporate brand equity over time.
Tools and Partners for Biotech Brand Strategy
Building effective biotech brand strategy requires a combination of internal commercial capability and specialized external partners. Core internal requirements include commercial leadership with both scientific fluency and business development experience, a market research function capable of conducting qualitative and quantitative studies with HCP, payer, and patient audiences, and a medical communications infrastructure capable of producing and distributing scientific content across congresses, publications, and digital channels. External partners typically include a life sciences brand strategy agency with therapeutic area expertise for positioning and messaging architecture; a KOL engagement and medical affairs partner for scientific communication and advisory board management; a market access and health economics firm for payer strategy and HEOR modeling; and a digital health communications partner for HCP and patient digital engagement. The selection of these partners is particularly important for first-time launchers: agencies with experience exclusively in large-pharma commercial models often bring approaches that are poorly calibrated for the resource constraints, speed requirements, and multi-stakeholder complexity that characterize emerging biotech commercial environments.
Common Misconceptions About Biotech Brand Strategy
The most damaging misconception is that superior science sells itself. In a competitive landscape where most therapeutic areas have multiple assets in development, clinical differentiation creates opportunity but does not automatically create commercial success, the brand narrative must translate the science into outcomes that each stakeholder audience cares about, in language that is credible, compliant, and accessible to non-specialist decision-makers. The second misconception is that brand strategy is primarily a marketing function. The most impactful commercial decisions in biotech, TPP design, indication selection, patient population definition, pricing strategy, market access planning, require commercial perspective integrated with clinical, regulatory, and business development thinking from the earliest stages of development. The third misconception is that pre-approval brand activity is primarily about awareness. While awareness is an outcome of pre-commercial investment, the primary objective is relationship and credibility building: with the KOLs whose scientific influence shapes prescribing behavior, with the payer decision-makers who determine coverage, and with the investors and partners whose capital enables continued development.
How G&Co. Can Help

G&Co. works with life sciences and biotech companies to build brand strategies that connect scientific differentiation to commercial value, at every stage of the development and commercialization journey. Through our Brand Strategy and Competitive & Competitor Intelligence capabilities, we help clinical-stage and commercial-stage biotech organizations define their brand narrative, structure their stakeholder communication architecture, and develop the go-to-market strategies that position assets for fundraising, partnership, and launch success. Our approach integrates brand strategy with competitive intelligence, market research, and experience design, ensuring that the positioning we develop is grounded in real stakeholder insight, differentiated in competitive context, and executable within the resource realities of the organizations we work with.
G&Co. is a minority business enterprise (MBE), as certified by the National Minority Supplier Development Council (NMSDC). If diversity inclusion is part of your supplier process, contact us, we may be a great fit for your enterprise.
Talk to us to clarify your biotech brand strategy and move forward with confidence.
Conclusion & Next Steps
Biotech brand strategy in 2026 is no longer a commercial luxury, it is a development-stage competitive necessity. The biotechs that are commanding premium valuations, attracting strategic partners, and achieving strong launch performance share a common characteristic: they invested in brand strategy and commercial readiness early, treating it as integral to the development program rather than a post-approval add-on. The scientific opportunity is not in question, the biotechnology market is growing at nearly 13% annually and the pipeline of innovative therapies has never been stronger. The question is whether the commercial infrastructure exists to translate that scientific innovation into market value.
The most important next step for any clinical-stage biotech is an honest assessment of commercial readiness: whether the brand narrative is clear and differentiated for each key audience, whether KOL and payer relationships are being built in advance of the data, and whether the go-to-market plan is designed for the commercial environment that will exist at launch rather than the environment that existed when the clinical program began. At G&Co., we've worked alongside life sciences clients to build exactly this foundation, from brand architecture through stakeholder engagement strategy and commercial launch planning. Still have questions? Reach out and let's solve them together.






%20(1).png)


