
Pfizer Case Study: AI-Enabled CX Transformation & Pharmaceutical Experience Strategy 2026
Strategic Overview
Pfizer operates across a $62.6 billion enterprise with more than 80,000 employees and one of the most complex pharmaceutical commercial environments in the world — yet its patient and HCP commercial experience was historically fragmented: drug brands communicating in silos, content creation cycles too slow to capitalize on launch windows, and no unified AI layer connecting commercial intelligence to patient engagement. The response has been a deliberate re-architecture of the entire pharmaceutical experience journey, deploying AI as connective tissue from drug discovery through to patient care — using Charlie (generative AI commercial platform), Golden Batch (manufacturing AI), PACT/AWS (discovery infrastructure), and an enterprise-wide Microsoft Copilot rollout as the four pillars of its CX transformation. The outcomes validate the approach: $1.5 billion Manufacturing Optimization Program on track by end of 2027; 16,000 scientist hours saved annually; $4.5 billion in total cost realignment savings through 2025, reinvested into the pipeline and patient experience infrastructure. The deeper lesson is strategic: AI-enabled CX transformation in pharma is not a technology project — it is a re-architecture of how a drug brand builds trust with patients and clinicians at every stage of the therapeutic journey, from discovery to ongoing care.

Let’s kickstart the conversation and design stuff people will love.

The Commercial Experience Problem at the Heart of Pharmaceutical AI
Pfizer enters 2026 as one of the most consequential pharmaceutical enterprises in the world, generating $62.6 billion in FY2025 revenue across dozens of therapeutic areas and global markets. But scale has historically been as much a liability as an advantage in pharmaceutical commercial experience. A drug brand manager at Pfizer's oncology division and a brand manager at its vaccines division could be operating with entirely different content workflows, different HCP segmentation models, different digital experience platforms, and different feedback loops from the patient journey, generating fragmented commercial experiences for clinicians and patients who increasingly expect the same level of personalization and responsiveness they receive from technology platforms in every other area of their lives.
This is the patient and HCP experience problem that Pfizer's AI transformation is fundamentally designed to solve, and it is the same problem that defines the strategic challenge for every major pharmaceutical enterprise in 2025 and 2026. The question is not whether to deploy AI in pharmaceutical commercial operations. It is how to architect AI so that it operates as the connective tissue between drug discovery, clinical evidence, brand communication, and ongoing patient engagement, rather than as a collection of isolated tools that accelerate individual tasks without improving the coherence of the overall experience journey. Pfizer's answer to that question, executed across four integrated layers of its enterprise, is the most instructive case study in pharmaceutical CX transformation available at this scale.
Why Pharmaceutical CX Transformation Is Different, and Why That Makes It More Valuable
Healthcare and pharmaceutical enterprises face a customer experience design challenge that no other industry confronts at equivalent complexity: the patient journey intersects simultaneously with clinical evidence, regulatory compliance, insurance authorization, provider communication, and ongoing therapeutic adherence, and every touchpoint in that journey is subject to a level of regulatory scrutiny that consumer brands never face. Promotional materials for a Pfizer drug must pass medical, legal, and regulatory review before any clinician or patient sees them. AI-generated adverse event summaries must meet FDA pharmacovigilance standards. Patient engagement content must comply with HIPAA. The compliance architecture is not a constraint on the experience, it is part of the experience, because patients and providers who do not trust a brand's regulatory integrity do not engage with its clinical messaging regardless of how well-designed it is.
This is why Pfizer's CX transformation case study is more consequential for pharmaceutical enterprise leaders than any consumer brand AI deployment. The organizations that solve AI-enabled experience design inside this compliance architecture, building systems that are simultaneously personalized, clinically credible, regulatory-compliant, and operationally fast, are building a commercial capability that cannot be replicated by less integrated competitors. Pfizer's Chief AI, Data & Analytics Officer Jeremy Forman, promoted to the role in January 2026, has explicitly framed the enterprise AI program not as a cost-efficiency initiative but as the infrastructure for building more meaningful and responsive relationships with patients and healthcare providers, a frame that positions AI as an experience enabler, not merely a productivity tool.
The Foundation: Building the Intelligence Infrastructure That Makes Better Experiences Possible
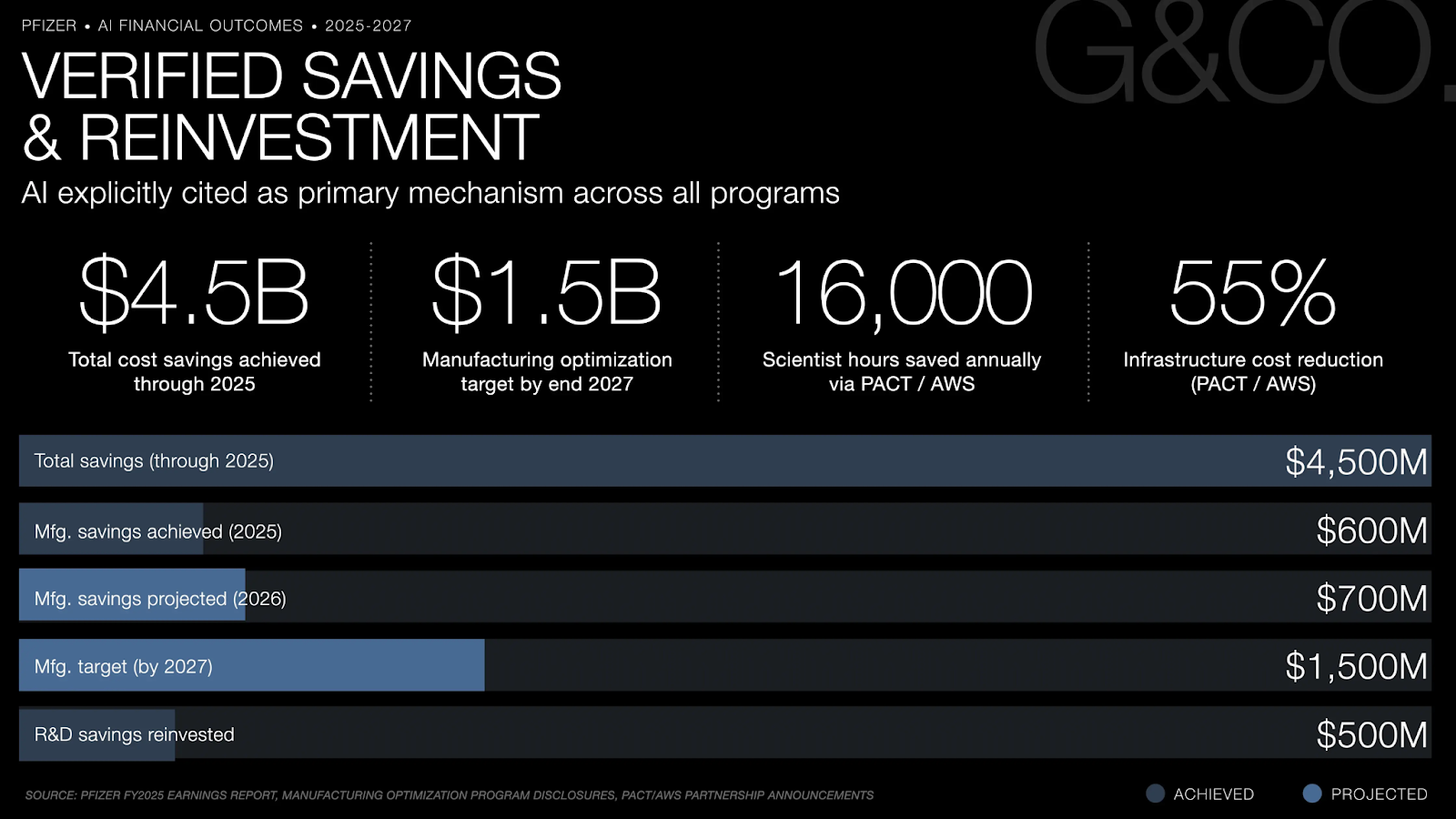
The patient and HCP experience improvements Pfizer is delivering in 2025 and 2026 are built on an AI infrastructure investment that began years earlier, a sequencing that most pharmaceutical enterprises attempting to accelerate commercial AI deployment underestimate. The Pfizer-Amazon Collaboration Team (PACT), structured around AWS cloud infrastructure, has executed 14 generative AI and machine learning projects that save scientists 16,000 hours annually while reducing infrastructure costs by 55%. These are not administrative savings, they represent research time redirected toward the clinical evidence generation that eventually becomes the content of HCP communications and patient education materials.
The partnership strategy Pfizer assembled between 2024 and 2026 tells the same story through a different lens. In the January 2026 collaboration with Boltz, PBC deploys biomolecular AI foundation models for drug design, with Pfizer retaining full ownership of resulting compounds. The Flagship Pioneering partnership, with its sixth collaboration announced in April 2025, targets novel selective inhibitors for autoimmune disease using the Logica AI platform, part of a broader umbrella agreement spanning up to 10 programs. The February 2025 Data4Cure agreement deploys LLMs and knowledge graphs for oncology analytics. The December 2025 Adaptive Biotechnologies deal, with up to $890 million in potential milestones, provides TCR-antigen data for AI training across disease areas. Taken together, these investments are building the proprietary clinical intelligence foundation that will power the next generation of personalized HCP communications and patient support programs. The experience layer is only as strong as the evidence layer beneath it.
Manufacturing Excellence as a Patient Experience Investment
Pfizer's Manufacturing Optimization Program, projecting $1.5 billion in net cost savings by end of 2027, with approximately $600 million achieved through 2025 and $0.7 billion projected for 2026, is typically framed as a cost efficiency story. From a patient experience lens, it is something more consequential: the infrastructure that ensures drug supply reliability, product quality consistency, and the financial headroom to invest in the patient support programs and digital health tools that define the post-prescription experience journey.
The Golden Batch initiative sits at the center of this manufacturing CX connection. By using AI to identify and replicate the optimal production conditions that generate the highest-quality pharmaceutical batches, and monitoring real-time manufacturing parameters against those optima, Pfizer is converting manufacturing from a process of managing variance to a process of replicating excellence. For patients on specialty therapeutics or complex biologics, manufacturing quality is not an abstract operational metric. It is the reliability of the medication they depend on. The $4.5 billion in total cost realignment savings Pfizer achieved through 2025, with AI explicitly cited as a primary mechanism, has been partially reinvested in the pipeline and patient experience infrastructure rather than returned exclusively to the bottom line, a deliberate decision that connects manufacturing efficiency directly to the quality of the patient journey downstream.
Clinical Development: Compressing the Time Between Evidence and Patient Access
One of the most consequential but least visible dimensions of pharmaceutical patient experience is the time between when clinical evidence exists and when patients can access the treatments that evidence supports. Regulatory submission timelines, pharmacovigilance processing delays, and documentation bottlenecks are not merely operational inefficiencies, they are patient experience failures, measured in months of delayed access for individuals with serious or rare diseases. Pfizer's AI deployment in clinical development directly targets this gap.
AI-generated tables, reports, and regulatory summary narratives for FDA filings compress the review preparation timeline for major drug submissions. Natural language processing applied to adverse event case processing accelerates pharmacovigilance signal detection, a compliance-critical function where faster signal identification protects patient safety and maintains the clinical credibility that underpins HCP trust. The PACT/AWS infrastructure compresses prototype-to-MVP timelines for clinical AI tools to as little as six weeks, allowing clinical operations teams to iterate rapidly on the digital tools that support trial participants and study sites. The $500 million in R&D savings achieved through the digital enablement component of the Cost Realignment Program, reinvested directly into the pipeline, represents Pfizer's explicit commitment to using AI efficiency in clinical operations as the funding mechanism for faster patient access to the next generation of therapies.
Charlie: The Generative AI Commercial Experience Platform That Changes What Pharma Marketing Can Do
Charlie, Pfizer's enterprise generative AI marketing platform, launched in February 2024 and named after co-founder Charles Pfizer, is the most visible expression of what AI-enabled pharmaceutical CX transformation looks like in practice, and the most instructive model for pharmaceutical enterprises asking what G&Co. means by AI Strategy & Enablement in a commercial context. Built in collaboration with Publicis Groupe using a customized version of Marcel's AI platform, Charlie serves as the connective layer between Pfizer's clinical evidence, brand strategy, regulatory compliance requirements, and the content that reaches healthcare providers and patients.
Charlie is used by hundreds of professionals in Pfizer's central marketing team and thousands across its brand teams, as well as by agency partners including Publicis and IPG. Its capabilities span content creation, editing, clinical fact-checking, legal review flagging, and media performance analytics, effectively collapsing what was previously a multi-week, multi-stakeholder content development and approval cycle into a system where AI generates compliant first drafts, flags regulatory risk in real time using a traffic-light color-coding system, and learns from approved content to continuously improve the quality of future outputs. The platform was trained on approved content organized by therapeutic category and product, combined with HCP and patient segmentation models that teach the system which messaging approaches resonate with each audience.
The strategic ambition Pfizer has articulated for Charlie goes further than operational efficiency. The goal, stated explicitly by Pfizer's VP of customer engagement, is to triple or quintuple content creation volume while simultaneously improving the precision and personalization of messaging for both healthcare providers and patients. This is the pharmaceutical CX transformation thesis made operational: not faster versions of the same generic content, but genuinely personalized clinical communications, tailored to an oncologist's specialty, a patient's disease stage, a payer's formulary position, delivered at a speed and scale that was previously impossible without AI. Charlie's NLP capabilities extend beyond marketing into internal research synthesis, competitive intelligence, and performance analytics, making it a commercial intelligence system that continuously improves the organization's understanding of how patients and HCPs actually engage with its brands.
The Patient and HCP Journey Reimagined: What AI-Enabled Pharmaceutical CX Actually Looks Like
The four layers of Pfizer's AI transformation, discovery, manufacturing, clinical development, and commercialization, only become a patient experience story when they are understood as a connected journey rather than isolated infrastructure investments. A patient diagnosed with a rare autoimmune disease in 2026 encounters Pfizer's AI investment at every stage of their therapeutic journey, whether or not they are aware of it. The drug they are prescribed was discovered using AI-assisted molecular modeling that identified a viable candidate years faster than traditional approaches. The manufacturing process that produced it was optimized using Golden Batch AI to minimize batch variability and maximize quality consistency. The clinical trial that generated the evidence their physician reviewed was supported by AI-generated regulatory documentation that accelerated FDA approval. And the patient support program their specialty pharmacist enrolled them in was personalized using Charlie's segmentation models to deliver the educational content and adherence communications most relevant to their specific disease stage and treatment history.
This is what CX transformation at scale means in a pharmaceutical context, and it is why the organizations that build it correctly create a patient and provider experience that is not replicable by competitors who have deployed AI in one or two functions without connecting them. The Microsoft Copilot rollout across tens of thousands of Pfizer employees, the 54-session AI Festival spanning seven countries, and the organizational investment in building baseline AI fluency across the entire workforce are not productivity programs. They are the human infrastructure investments that ensure every person who touches the patient or HCP experience, from the field medical liaison to the patient services coordinator to the brand manager, has the tools and capability to deliver a more intelligent, more responsive, more empathetic interaction than was possible before.

Governing AI in a Regulated Experience Environment
The compliance architecture that governs Pfizer's AI deployment is not a constraint on its patient experience ambitions, it is the foundation of clinical trust that makes those ambitions commercially viable. Patients and healthcare providers do not engage deeply with pharmaceutical brands whose AI systems they do not trust. VP of Compliance for AI Lucy Muzzy's personal authorship of Pfizer's first AI governance policy and its Three Principles of Responsibility, empowering humans, respecting privacy, and maintaining transparency, are not legal formalities. They are the experience design commitments that make it possible for an oncologist to trust that Charlie's AI-generated clinical summary is accurately sourced, for a patient to trust that their data is being used to personalize their care rather than to surveil their behavior, and for a payer to trust that Pfizer's AI-assisted outcomes analytics are methodologically sound.
The AI Council, reporting through Chief AI, Data & Analytics Officer Jeremy Forman, ensures that governance decisions and technology decisions are made simultaneously rather than sequentially. This is the organizational design pattern that pharmaceutical enterprises must replicate to build AI-enabled experience systems that are both clinically credible and commercially effective: compliance as a design input, not a deployment gate. Pfizer's governance architecture is itself a patient experience asset, because the trust it generates is the foundation on which every other element of the experience journey depends.
What Pfizer's CX Transformation Reveals for Pharmaceutical Enterprise Leaders
The repeatable pattern Pfizer's transformation crystallizes is this: pharmaceutical enterprises that deploy AI as an experience design system, connecting clinical evidence to commercial communication to patient support through a unified AI layer, create a patient and HCP experience that is structurally more valuable than any individual tool can produce. The Charlie platform is valuable because it is trained on Pfizer's proprietary clinical evidence base. The Golden Batch manufacturing system is valuable because its quality outputs underpin the clinical credibility that Charlie communicates. The PACT/AWS discovery infrastructure is valuable because it generates the molecular insights that eventually become the treatment options patients receive. Each layer depends on and amplifies the others.
For pharmaceutical enterprise leaders evaluating where to invest in AI-enabled experience transformation, the Pfizer case study offers two clear sequencing principles. First: the experience layer requires the evidence layer. Organizations that invest in AI-enabled marketing without investing in the clinical data infrastructure that makes personalized, credible communication possible will produce sophisticated-looking content that clinicians and patients do not trust. Second: compliance architecture must precede scale. Organizations that attempt to scale AI-enabled patient and HCP engagement without building the governance infrastructure that makes those deployments regulatory-compliant will produce experiences that expose the organization to material risk precisely at the moment when they are generating the most patient engagement.
Executive Takeaways
- AI-enabled pharmaceutical CX transformation requires a connected architecture spanning the full patient journey, from discovery to commercialization, because the experience is only as credible as the clinical evidence and operational reliability that underpin it.
- The Charlie platform demonstrates that generative AI for pharmaceutical commercial experience must be trained on proprietary clinical and brand content to deliver the personalization and compliance accuracy that HCPs and patients require, generic LLM deployments cannot achieve this without the underlying data architecture.
- Compliance governance must be designed as an experience enabler, not a deployment gate, Pfizer's Three Principles of Responsibility for AI are a trust-building commitment to patients and clinicians as much as they are an internal policy framework.
- Manufacturing and supply chain AI are patient experience investments, not merely efficiency programs, drug quality consistency and supply reliability are the foundation of therapeutic trust that no amount of commercial AI can compensate for if they fail.
- Enterprise AI fluency across the entire commercial organization, not just the data science and technology functions, is the human infrastructure prerequisite for delivering AI-enabled patient and HCP experiences that are consistently empathetic, responsive, and clinically credible at scale.
Why This Matters Now for Pharmaceutical and Life Sciences Enterprises
The structural conditions driving Pfizer's AI-enabled CX transformation, patent cliff pressure requiring faster commercial velocity, patient expectations shaped by consumer technology platforms, HCP communication preferences shifting toward personalized digital engagement, and regulatory environments demanding greater transparency in AI-assisted clinical communications, are not unique to Pfizer. They define the operating reality of every major pharmaceutical and life sciences enterprise in 2025 and 2026. The organizations investing in AI-enabled experience architecture now are establishing patient and HCP relationships that compound in value over time: better data generates better personalization, which generates deeper engagement, which generates richer data.
The window for pharmaceutical enterprises to establish this compounding advantage is not unlimited. The organizations that deploy AI as an integrated experience system first, connecting clinical evidence to commercial communication to patient support through a governed, scalable AI architecture, will build patient and provider relationships that are structurally harder for competitors to displace with each passing year. Pfizer's transformation, executed under simultaneous revenue pressure and regulatory constraint at $62.6 billion in annual revenue, is proof that this architecture is buildable at the highest level of pharmaceutical complexity. The question for every other enterprise in space is not whether to build it, it is how to build it fast enough to matter.
Conclusion
Pfizer's AI-enabled CX transformation crystallizes the central challenge facing every pharmaceutical and life sciences enterprise in 2026: the organizations that will lead patient and HCP experience over the next decade are not the ones with the most sophisticated individual AI tools, they are the ones that connect those tools into a coherent experience architecture that earns clinical trust, delivers genuine personalization, and operates reliably inside the compliance environment that makes pharmaceutical engagement meaningful rather than merely transactional.
The enduring lesson is not about artificial intelligence. It is about what healthcare should be, an experience built around real people facing real challenges, designed with empathy and delivered with clinical integrity. Pfizer's transformation, playing out at $62.6 billion in annual revenue across dozens of therapeutic areas and global markets, demonstrates that this level of experience coherence is achievable at pharmaceutical enterprise scale. For life sciences and pharmaceutical leaders asking whether their organization can build what Pfizer has built, the answer is yes. The architecture is proven. The sequencing is documented. The compliance framework is established. What remains is the organizational commitment to treat AI not as a productivity tool, but as the infrastructure for building the most trusted, most responsive, most human pharmaceutical experience in the market.
Ready to transform your pharmaceutical commercial experience?
Submit an inquiry to G & Co. on our contact page or click on the blue "Click to Contact Us" button on the bottom right corner of your screen for your convenience. We look forward to hearing from you.
Frequently Asked Questions
How is Pfizer using AI to improve patient and HCP experience?
Pfizer's AI deployment spans the full pharmaceutical experience journey: the PACT/AWS discovery infrastructure generates the clinical evidence that underpins credible HCP communications; Golden Batch manufacturing AI ensures the drug quality consistency that defines the patient's therapeutic experience; Charlie's generative AI platform personalizes and accelerates compliant content delivery to healthcare providers and patients; and an enterprise-wide Microsoft Copilot rollout ensures that every person touching the patient or HCP experience has AI capability embedded in their workflow.
How is Pfizer using AI to improve patient and HCP experience?
Pfizer's AI deployment spans the full pharmaceutical experience journey: the PACT/AWS discovery infrastructure generates the clinical evidence that underpins credible HCP communications; Golden Batch manufacturing AI ensures the drug quality consistency that defines the patient's therapeutic experience; Charlie's generative AI platform personalizes and accelerates compliant content delivery to healthcare providers and patients; and an enterprise-wide Microsoft Copilot rollout ensures that every person touching the patient or HCP experience has AI capability embedded in their workflow.
How does Pfizer govern AI in a regulated pharmaceutical environment?
Pfizer built compliance governance into the organizational design of its AI program through its Three Principles of Responsibility for AI — empowering humans, respecting privacy, and maintaining transparency — authored by VP of Compliance Lucy Muzzy. The AI Council, reporting through Chief AI, Data & Analytics Officer Jeremy Forman, ensures that technology and compliance decisions are made simultaneously. This governance architecture is both a regulatory requirement and a patient and HCP trust-building commitment.
What are the verified financial outcomes of Pfizer's AI-enabled transformation?
Pfizer achieved approximately $4.5 billion in total net cost savings through 2025, with AI explicitly cited as a primary mechanism. The Manufacturing Optimization Program is on track to deliver $1.5 billion in savings by the end of 2027, with $0.7 billion projected for 2026. The PACT/AWS collaboration saves 16,000 scientist hours annually and reduces infrastructure costs by 55%. $500 million in R&D savings have been reinvested directly into the pipeline.
What can pharmaceutical enterprise leaders learn from the Pfizer AI case study?
The primary lesson is architectural: pharmaceutical enterprises that deploy AI as an integrated experience system — connecting clinical evidence infrastructure to commercial communication to patient support through a governed AI layer — create patient and HCP relationships that compound in value and are structurally difficult for competitors to replicate. Organizations that deploy AI in isolated functions without connecting them produce speed without trust, and efficiency without the clinical credibility that determines whether patients and providers actually engage.






%20(1).png)




